2-chloroethanol News
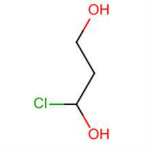
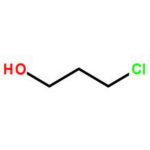
2-chloroethanol
-

- Cas No.:107-07-3
- Appearance:Colorless liquid
- Molecular Formula:C2H5ClO
- ProductionCapacity:
- Inquiry
Detailed information
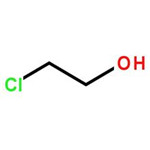
2-Chloroethanol Chemical Structure
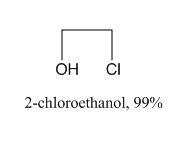
Formula C2H5ClO
Molecular weight 80.52
CAS No 107-07-3
Appearance Colorless liquid
Assay (GC, area%) ≥ 99%
Acid value(Calculation based HCl) ≤ 0.05%
Water (according to Karl Fischer) ≤ 0.10%
Density (at 20°C, g/ml) 1.200 - 1.203
Package: Special drum of hazardous chemicals , Net weight 200kg/drum.
2-Chloroethanol is an chemical compound with the formula HOCH2CH2Cl and the implest chlorohydrin. This colorless liquid has a pleasant ether-like odor. It is miscible with water. The molecule is bifunctional, consisting of both an alkyl chloride and an alcohol functional groups.
2-chloroethanol Uses
2-Chloroethanol is widely used in the synthesis of pharmaceutical intermediates, pesticide intermediates and fine chemicals. It is a good organic solvent, and is also used in printing and electronic products for cleaning.
2-chloroethanol is a building block in the production of pharmaceuticals, biocides and plasticizers. It is also used for manufacture of thiodiglycol. It is a solvent for cellulose acetate and ethyl cellulose, textile printing dyes, extraction of pine lignin, in dewaxing, refining of rosin, and the cleaning of machines. Several dyes are prepared by the alkylation of aniline derivatives with chloroethanol.
2-Chloroethanol Synthesis and applications
2-Chloroethanol is produced by treating ethylene with hypochlorous acid:
![]()
2-Chloroethanol was once produced on a large scale as a precursor to ethylene oxide:
![]()
This application has been supplanted by the greener direct oxidation of ethylene. Otherwise chloroethanol is used in a number of specialized applications.Several dyes are prepared by the alkylation of aniline derivatives with chloroethanol. It is a building block in the production of pharmaceuticals, biocides and plasticizers. It is also used for manufacture of thiodiglycol. It is a solvent for cellulose acetate and ethyl cellulose, textile printing dyes, in dewaxing, refining of rosin, extraction of pine lignin, and the cleaning of machines.
2-chloroethanol is synthesized by three step
first step: the direct reaction of ethylene oxide and hydrochloric acid
second step: removing the water by the way of the co-boiling
third step: distillation
2-chloroethanol Toxicity Data With Reference
1. skn-rbt 200 mg/2H MLD
TXAPA9 Toxicology and Applied Pharmacology. 16 (1970),382.
2. eye-rbt 2 mg SEV
AJOPAA American Journal of Ophthalmology. 29 (1946),1363.
3. eye-rbt 33 mg MOD
TXAPA9 Toxicology and Applied Pharmacology. 16 (1970),382.
4. eye-rbt 9 mg/6H MOD
BUYRAI Bulletin of Parenteral Drug Association. 31 (1977),25.
5. sln-asn 74,500 μmol/L
MUREAV Mutation Research. 138 (1984),33.
6. oms-rat:lvr 12 g/L
JACTDZ Journal of the American College of Toxicology. 1 (3)(1982),37.
7. ihl-man LCLo:305 ppm/2H
JIHTAB Journal of Industrial Hygiene and Toxicology. 26 (1944),277.
8. orl-rat LD50:71 mg/kg
HYSAAV Hygiene and Sanitation: English Translation of Gigiena Sanitariya. 36 (1971),376.
9. ihl-rat LC50:290 mg/m3
HYSAAV Hygiene and Sanitation: English Translation of Gigiena Sanitariya. 26 (1971),376.
10. ipr-rat LD50:58 mg/kg
TXAPA9 Toxicology and Applied Pharmacology. 21 (1972),454.
RTECS :KK0875000
2-chloroethanol is toxic with an LD50 of 89 mg/kg in rats.
2-chloroethanol Consensus Reports
NTP Carcinogenesis Studies (dermal); No Evidence: mouse, rat NTPTR* National Toxicology Program Technical Report Series. (Research Triangle Park, NC 27709) No. NTP-TR-275 ,1985. ; Reported in EPA TSCA Inventory. EPA Genetic Toxicology Program. EPA Extremely Hazardous Substances List.
2-chloroethanol Safety Profile
A poison by ingestion, inhalation, skin contact, intraperitoneal, intravenous, and subcutaneous routes. Moderately toxic to humans by inhalation. It can affect the nervous system, liver, spleen, and lungs. An experimental teratogen. Mutation data reported. A severe eye and mild skin irritant. Flammable liquid when exposed to heat, flame, or oxidizers. To fight fire, use alcohol foam, CO2, dry chemical. Violent reaction with chlorosulfonic acid, ethylene diamine, sodium hydroxide. Reacts with water or steam to produce toxic and corrosive fumes. Potentially violent reaction with oxidizing materials. When heated to decomposition it emits highly toxic fumes of Cl and phosgene. See also CHLORINATED HYDROCARBONS, ALIPHATIC.
Like most organochlorine compounds, 2-chloroethanol combusts to yield hydrogen chloride and phosgene.
Hazard Codes: T+,T,F
Risk Statements : 26/27/28-39/23/24/25-11
Safety Statements : 28-45-7/9-28A-36/37-16
RIDADR :UN 1135 6.1/PG 1
WGK Germany 3
2-Chloroethanol is toxic with an LD50 of 89 mg/kg in rats. Like most organochlorine compounds, chloroethanol combusts to yield hydrogen chloride and phosgene.
In regards to dermal exposure to 2-chloroethanol, the Occupational Safety and Health Administration has set a permissible exposure limit of 5 ppm (16 mg/m3) over an eight-hour time-weighted average, while the National Institute for Occupational Safety and Health has a more protective recommended exposure limit of a 1 ppm (3 mg/m3) exposure ceiling.
2-chloroethanol Standards and Recommendations
OSHA PEL: CL 1 ppm (skin)
ACGIH TLV: CL 1 ppm (skin); Not Classifiable as a Human Carcinogen
DFG MAK: 1 ppm (3.3 mg/m2)
DOT Classification: 6.1; Label: Poison
2-chloroethanol Analytical Methods
For occupational chemical analysis use NIOSH: 2-chloroethanol, 2513.
2-chloroethanol Specification
Other names of 2-chloroethanol :2-chloroethyl alcohol, 2-chloroethanol, 2-chloro-1-ethanol, 2-monochloroethanol,glycol chlorohydrin, 2-hydroxyethyl chloride, β-hydroxyethyl chloride, β-chloroethanol,chloroethanol,ethylchlorhydrin, δ-chloroethanol, 2-chloroethanol, glycol monochlorohydrin.
the following control is recommended to 2-chloroethanol
where possible, automatically pump liquid 2-chloroethanol from drums or other storage containers to process containers.good work practices can help to reduce hazardous
exposures.
the following work practices are recommended to 2-chloroethanol
workers whose clothing has been contaminated by 2-chloroethanol should change into clean clothing promptly.
contaminated work clothes should be laundered by individuals who have been informed of the hazards of exposure to 2-chloroethanol.
eye wash fountains should be provided in the immediate work area for emergency use.
if there is the possibility of skin exposure, emergency shower facilities should be provided.
on skin contact with ethylene 2-chloroethanol,immediately wash or shower to remove the chemical. at the end of the workshift, wash any areas of the body that may have contacted 2-chloroethanol, whether or not known skin contact has occurred.
do not eat. smoke. or drink where 2-chloroethanol is handled, processed. or stored, since the chemical can be swallowed. wash hands carefully before eating, drinking, smoking, or using the toilet.
2-chloroethanol handling and storage
prior to working with 2-chloroethanol you should be trained on its proper handling and storage.
ethylene 2-chloroethanol reacts violently with chlorosulfonic acid; ethylene diamine;sodium hydroxide and oxidizing agents(such as perchlorates. peroxides,permanganates. chlorates. nitrates,chlorine. bromine and fluorine).
2-chloroethanol is not compatible with strong bases (such as potassium hydroxide); strong acids (such as hydrochloric, sulfuric and nitric); and alkaline metals.
store in tightly closed containers in a cool. well-ventilated area away from water and steam as corrosive gases are produced.
sources of ignition. such as smoking and open flames, are prohibited where 2-chloroethanol is used, handled. or stored in a manner that could create a potential fire or explosion hazard.
2-Chloro-1-phenylethanol
Molecular FormulaC8H9ClO
Average mass156.609 Da
Monoisotopic mass156.034195 Da
ChemSpider ID83862
Names and identifiers
1674-30-2 [RN]
216-816-7 [EINECS]
2-Chlor-1-phenylethanol [German] [ACD/IUPAC Name]
2-Chloro-1-phenylethanol [ACD/IUPAC Name]
2-Chloro-1-phényléthanol [French] [ACD/IUPAC Name]
907322 [Beilstein]
Benzenemethanol, α-(chloromethyl)- [ACD/Index Name]
Benzyl alcohol, α-(chloromethyl)-
QYR&1G [WLN]
(1S)-2-Chloro-1-phenylethanol [ACD/IUPAC Name]
(Chloromethyl)phenyl carbinol
(R)-(-)-2-CHLORO-1-PHENYLETHANOL CHIPROS?
2-Chloro-1-phenylethan-1-ol
MFCD01075658 [MDL number]
Styrene chlorohydrin
Styrene β-chlorohydrin
α-(Chloromethyl)benzyl alcohol
α-(Chloromethyl)benzyl alcohol
2-Chloro-1-(4-chlorophenyl)ethanol
Molecular FormulaC8H8Cl2O
Average mass191.055 Da
Monoisotopic mass189.995224 Da
ChemSpider ID99481
Names and identifiers
228-954-5 [EINECS]
2-Chlor-1-(4-chlorphenyl)ethanol [German] [ACD/IUPAC Name]
2-Chloro-1-(4-chlorophenyl)ethanol [ACD/IUPAC Name]
2-Chloro-1-(4-chlorophényl)éthanol [French] [ACD/IUPAC Name]
Benzenemethanol, 4-chloro-α-(chloromethyl)- [ACD/Index Name]
2-chloro-1-(4-chlorophenyl)-ethanol
2-Chloro-1-(4-chloro-phenyl)-ethanol
4-chloro-α-(chloromethyl)benzyl alcohol
6378-66-1 [RN]
MFCD00462592
2-Chloro-1-(2,4-dichlorophenyl)ethanol
Molecular FormulaC8H7Cl3O
Average mass225.500 Da
Monoisotopic mass223.956253 Da
ChemSpider ID85152
Names and identifiers
13692-14-3 [RN]
237-206-7 [EINECS]
2-Chlor-1-(2,4-dichlorphenyl)ethanol [German] [ACD/IUPAC Name]
2-Chloro-1-(2,4-dichlorophenyl)ethanol [ACD/IUPAC Name]
2-Chloro-1-(2,4-dichlorophényl)éthanol [French] [ACD/IUPAC Name]
Benzenemethanol, 2,4-dichloro-α-(chloromethyl)-
Benzenemethanol, 2,4-dichloro-α-(chloromethyl)- [ACD/Index Name]
Benzyl alcohol, 2,4-dichloro-α-(chloromethyl)-
[13692-14-3]
1-(2,4-dichlorophenyl)-2-chloroethan-1-ol
2,4-Dichloro-α-(chloromethyl)benzyl alcohol
2,4-Dichloro-α-(chloromethyl)benzyl alcohol
2,4-Dichloro-α-(chloromethyl)benzylalcohol?
2,4-Dichloro-α-chloromethylbenzenemethanol
?±-(chloromethyl)-2,4-dichlorobenzyl alcohol
MFCD00000950 [MDL number]
α-(2,4-Dichlorophenyl)-β-chloroethanol
α-(chloromethyl)-2,4-dichlorobenzyl alcohol
α-(Chloromethyl)-2,4-dichlorobenzyl alcohol
α-(CHLOROMETHYL)-2,4-DICHLOROBENZYLALCOHOL
Efficient Synthesis of (R)-2-Chloro-1-(3-chlorophenyl)ethanol by Permeabilized Whole Cells of Candida ontarioensis
(R)-2-Chloro-1-(3-chlorophenyl)ethanol is a key pharmaceutical intermediate in the synthesis of 3-adrenoceptor receptor (3-AR) agonists. The asymmetric reduction of 2-chloro-1-(3-chlorophenyl)ethanone to (R)-2-chloro-1-(3-chlorophenyl)ethanol catalyzed by resting cells of Candida ontarioensis was studied. At a substrate concentration of 10 g/L, the microbial cells showed excellent catalytic activity under the optimum reaction conditions, giving the product in 99.9% ee and 99.0% yield. After cetyltrimethylammonium bromide-pretreatment, the activity of permeabilized Candida ontarioensis cells was increased by more than 2-fold and the product could be produced over the significantly shortened reaction period of 24 h in 99.9% ee and 97.5% yield at a substrate concentration of 30 g/L. This work provides a practical approach for the efficient preparation of the important chiral intermediate (R)-2-chloro-1-(3-chlorophenyl) ethanol.
Kinetic Resolution of 2-Chloro-1-(3,4-dichlorophenyl)ethanol by Lipase-Catalyzed Transesterification
Kinetic resolution of racemic 2-chloro-1-(3,4-dichlorophenyl)ethanol was performed by free Alcaligene sp. lipase-catalyzed irreversible transesterification affording the (R)-isomer with ≧ 95% ee and the (S)-isomer with ≧ 90% ee. The activity of lipase Alcaligene sp. strongly depends on the basicity of the reaction system, and an organic base such as triethylamine can enhance the activity of the lipase and enantioselectivity markedly.
2-chloro-1-cyclohexyl-ethanol
SMILES String: C1CCC(CC1)C(CCl)O
Also know as: 2-chloro-1-cyclohexylethanol, 2-chloro-1-cyclohexylethanol, 2-chloro-1-cyclohexyl-ethanol, 2-chloro-1-cyclohexyl-ethanol
InChI=1/C8H15ClO/c9-6-8(10)7-4-2-1-3-5-7/h7-8,10H,1-6H2
Molecular Formula: C8H15ClO
Reactions where this compound is a product:
(Alkene Halogenation)
(Halohydrin formation from alkenes)
Reactions where this compound is a reactant:
(Ester Formation from Carboxylic Acid)
(Nucleophilic Acylation)
(Sn2)
(Enolate Substitution)
Preparation of Chiral 2-Chloro-1-(2,4-difluorophenyl)ethanol by Lipase-Catalyzed Resolution and Synthesis of Optically Active Antifungal Drug
Key chiral intermediates (R)- and (S)-2-chloro-1-(2,4-difluorophenyl)ethanols were prepared by kinetic resolution of the racemic mixture with Pseudomonas stutzeri lipase-catalyzed irreversible transesterification yielding (R)-alcohol and (S)-acetate, the (R)-alcohol with 95.8% ee and (S)-alcohol with 94.5% ee were obtained. And four optically active 2,4-difluoro-1-[1-(p-chlorobenzyloxy)-2-(1-imidazolyl)]ethyl- benzene nitric acid compounds possessing antifungal activity were synthesized. The results of antifungal tests in vitro by a method of filter paper disk diffusion showed that (R)-5a and (R)-5b had excellent antifungal activity against various candida species, and equal to fluconazole. Especially, 5a, 5b and their two optically active isomers have excellent antifungal efficacy against Aspergillus fumigatus that is resistant to fluconazole, moreover their (R)-isomers are more potent than their (S)-isomers and the racemate.
2-chloroethanol images










 < /a>
< /a>